Background: Light chain (AL) amyloidosis is a rare disease in which deposits of insoluble amyloid derived from immunoglobulin light chains accumulate and cause dysfunction of organs including the heart and kidneys. In order to reverse organ damage and improve overall survival, treatment must provide rapid, deep, and sustained hematologic responses. Although therapies approved for the treatment of multiple myeloma have demonstrated some efficacy, there are currently no health authority-approved treatments for AL amyloidosis. A retrospective observational study was undertaken to characterize patients and describe treatment patterns in patients with AL amyloidosis from real-world data in the US.
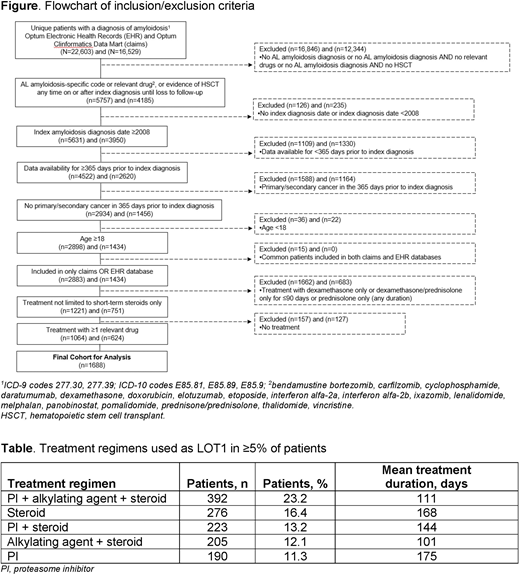
Methods: Data were extracted from Optum Electronic Health Records (EHR) and Optum Clinformatics Data Mart (claims) databases. Eligible patients had (1) a diagnosis of AL amyloidosis (ICD-10 code E85.81) on or after January 1, 2008 or (2) a diagnosis of amyloidosis (ICD-9 codes 277.30, 277.39; ICD-10 codes E85.89, E85.9) on or after January 1, 2008 and at least one line of therapy (LOT) comprising one or more of the following treatments: bendamustine bortezomib, carfilzomib, cyclophosphamide, daratumumab, dexamethasone, doxorubicin, elotuzumab, etoposide, interferon alfa-2a, interferon alfa-2b, ixazomib, lenalidomide, melphalan, panobinostat, pomalidomide, prednisone/prednisolone, thalidomide, vincristine, or autologous stem cell transplant. Patients were required to have continuous medical enrollment during the 365 days prior to the index amyloidosis diagnosis date (claims data) or have first active date at least 365 days prior to index amyloidosis date (EHR data), must not have had prior cancer in the 365-day period prior to the index amyloidosis diagnosis date, and must have been age ≥18 years at time of amyloidosis diagnosis. Patients treated with dexamethasone only or dexamethasone and prednisolone only for ≤90 days and those treated with prednisone/prednisolone only irrespective of duration were excluded.
LOTs were defined by a start date, an end date, and a distinct regimen made up of ≥1 drugs. Patients may have multiple sequential LOTs during the available follow-up period. Comorbidities were scored according to the Charlson Comorbidity Index (CCI). A score of 0 reflects no comorbidities; higher scores reflect increased mortality risk and the maximum score is 24.
Descriptive statistics are provided for patient characteristics at index diagnosis date for patients who had ≥1 LOT, distribution of regimens in each LOT, treatment attrition, and duration of treatment in each line of therapy and by regimen.
Results: Data from 1688 patients (Optum claims, n=624; Optum EHR, n=1064) were included in the analysis (Figure). Median age at diagnosis was 67 years and 55.7% of patients were male. Almost 70% of patients had an index diagnosis date between 2013 and 2019. The median CCI score at index diagnosis date (based on comorbidities in the 365-day pre-index diagnosis period) was 1 and 10.9% of patients had a stem cell transplant on or after the index diagnosis date. The three most common comorbidities were hypertension (50.8%), renal failure (26.2%), and congestive heart failure (24.6%). Median follow-up period was 28 months. Treatment information for regimens used by ≥5% of patients for LOT1 is summarized in the Table. The most common treatment regimen for LOT1 was a proteasome inhibitor (PI), an alkylating agent, and a steroid (n=392, 23.2%); this combination was used for an average of 111 days. Of patients treated with this combination, the most common regimen was bortezomib, cyclophosphamide, and dexamethasone (VCd), used by 358 patients. Steroid only (dexamethasone for ≥90 days) regimens accounted for 16.4% and a PI ± steroid for 13.2% of LOT1. A limitation of the study is that assumptions were used to classify AL amyloidosis vs other types of amyloidosis.
Conclusions: VCd is the most commonly used regimen in patients with newly-diagnosed AL amyloidosis. This is consistent with clinical guidelines and real world data from Europe (Palladini et al, EHA 2020), supporting this regimen as the current standard of care for treatment of patients with newly-diagnosed AL amyloidosis.
Dispenzieri:Celgene: Research Funding; Pfizer: Research Funding; Alnylam: Research Funding; Intellia: Research Funding; Janssen: Research Funding; Takeda: Research Funding. Zonder:Intellia, Amgen, Takeda, Janssen, Regeneron, Alnylam, Caelum, Oncopeptides: Consultancy; BMS, Celgene: Research Funding. Hoffman:Loxo: Current equity holder in publicly-traded company; Celgene: Honoraria, Speakers Bureau. Wong:Sanofi: Membership on an entity's Board of Directors or advisory committees; Janssen: Research Funding; Fortis: Research Funding; Roche: Research Funding; GSK: Research Funding; Amgen: Consultancy; Bristol Myers Squibb: Research Funding. Liedtke:Adaptive: Membership on an entity's Board of Directors or advisory committees; Caelum: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; GSK: Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Pfizer: Honoraria. Abonour:BMS: Consultancy, Research Funding; Takeda: Consultancy; Janssen: Honoraria, Research Funding; Celgene: Consultancy. D'Souza:Amgen, Merck, TenoBio: Research Funding; Akcea, Imbrium, Janssen, Pfizer: Consultancy. Lee:Janssen: Current Employment. Nair:Janssen: Current Employment. Potluri:SmartAnalyst Inc.: Current Employment. Weiss:Janssen: Current Employment, Current equity holder in publicly-traded company. Vermeulen:Janssen: Current Employment, Current equity holder in publicly-traded company. Lam:Janssen: Current Employment. Mehra:Janssen: Current Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal